Our Science
Creating IMPAKT
ImmVue is developing a new class of immunotherapies built on allosteric small molecule drugs, discovered using AI-driven computational chemistry.
Our platform, IMPAKT™ (Immune Modulating Precision Allosteric Kinase Therapy), focuses on the intracellular signaling pathways that control immune cell activation. By precisely tuning these signals inside immune cells, ImmVue aims to create therapies that can strengthen or restrain immune responses.
ITK: The Central Regulator of T-cell Activation

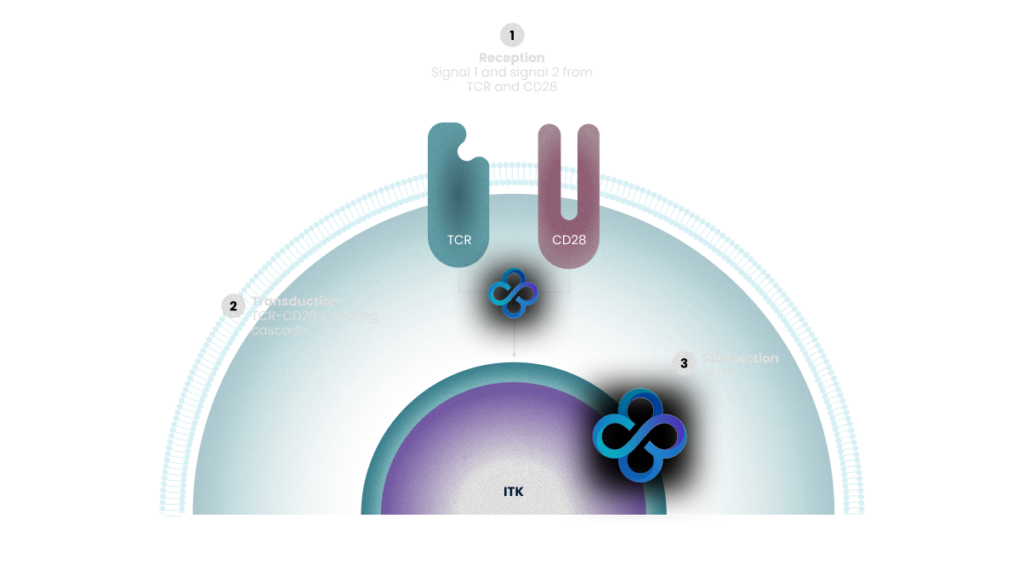
At the center of ImmVue’s approach is ITK (IL-2 Inducible T-cell Kinase), a key signaling protein that controls how T-cells respond to threats. ITK acts as a signal regulator inside T-cells, controlling the strength of the immune response. Rather than functioning as a simple on/off switch, ITK behaves more like a rheostat, allowing the immune response to be dialed up or down depending on the signals the cell receives.
By precisely modulating ITK activity, ImmVue aims to fine-tune immune responses rather than forcing them into extremes.
Where T-cell Signals Converge
T-cells become activated when they receive signals from two important receptors on their surface
Inside the cell, ITK integrates signals from both the TCR and CD28 pathways, helping to determine the quality and extent of T-cell response
ITK sitting at this critical junction of immune signaling represents a powerful target for precisely controlling immune activity
T-cell Receptor (TCR)
Recognizes specific antigens from infected or cancerous cells
CD28 Costimulatory Receptor
Provides an additional signal confirming that the immune response should proceed
Allosteric Binding: Our Pathway to Control Immune Proteins
Allosteric binding occurs when a drug attaches to a regulatory site on a protein that is separate from its primary active site. Rather than blocking the protein directly, binding at this site changes the protein’s shape and behavior, allowing its activity to be modulated
ImmVue has identified allosteric binding sites on immune kinases, such as ITK, using advanced computational chemistry and structural biology tools. Unlike traditional kinase drugs that target the highly conserved active site of enzymes, allosteric drugs bind to distinct regulatory regions of the protein

Greater precision in modulating immune signaling
Tunable control of protein activity rather than complete shutdown

Reduced off-target toxicity compared with traditional kinase inhibitors
